Everyday Concepts-Part 4
Radiation
Firefighters are often provided with an oversimplified explanation of fundamental scientific concepts related to fire behavior. This is done with the intent to make training manuals and texts understandable and to focus on the information that firefighters “must know”. However, a tremendous opportunity to develop the ability to make sense of fire dynamics and the impact of tactical operations is lost in the process. This series of posts continues to explore ways of building a scaffold to allow firefighters to develop a deeper understanding of firefighting as science.
Electromagnetic Radiation
The term radiation is used to describe many different things ranging from visible light, infrared light, and ionizing radiation such as x or gamma rays. Each of these is an example of radiation as an electromagnetic wave produced by the motion of electrically charged particles. Electromagnetic radiation can travel through empty space and air. Radiation can also penetrate through other materials depending on the characteristics of the material and the radiation’s energy. Some ionizing radiation is in the form of particles (rather than waves), but that is outside the scope of our examination of radiation as a mechanism of heat transfer.
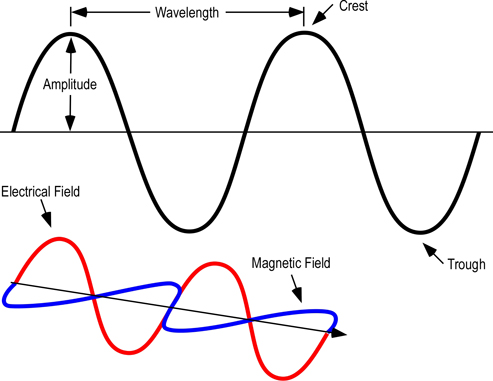
As illustrated in Figure 1, electromagnetic waves can be described in terms of their wavelength, amplitude, frequency, and energy.
Most of the electromagnetic spectrum cannot be detected by the human eye. While the electromagnetic spectrum includes radiation in a broad range of wavelengths, those of most interest in the study of fire behavior are categorized as infrared
Figure 1. Electromagnetic Wave
From longest to shortest wavelengths, the spectrum is usually divided into the following sections: radio, microwave, infrared, visible, ultraviolet, x-ray, and gamma-ray radiation. Humans can only see a narrow band of visible light, which is a small fraction of the electromagnetic spectrum. We perceive this radiation as the colors of the rainbow ranging from red to violet, with reds having longer wavelengths and violet having shorter wavelengths
Thermal radiation is electromagnetic radiation emitted from the surface of an object which is due to the object’s temperature. Any material that is above absolute zero gives off some radiant energy. Thermal radiation is generated when heat from the movement of charged particles within atoms is converted to electromagnetic radiation.
Figure 2. Electromagnetic Spectrum
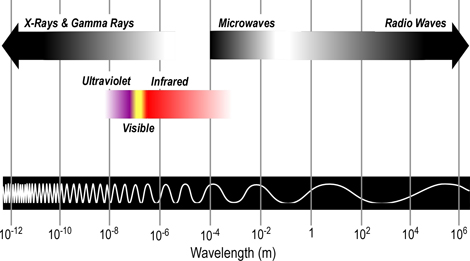
Figure 3. Planck’s Curve
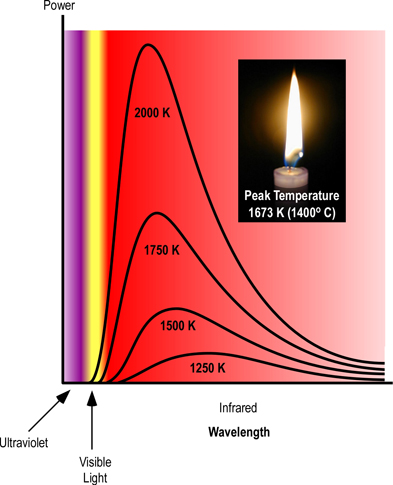
Thermal radiation occurs at a wide range of frequencies. However, as illustrated in Figure 2, the power emitted at each wavelength is dependent on temperature, with the main frequency and power of emitted radiation increasing as temperature increases. This can be observed when color changes from red, to yellow, and then white as an object is heated. While color change is visible, most of the radiant energy is still in the infrared spectrum.
Electromagnetic waves of any frequency will heat surfaces that absorb them. However, temperatures of hot surfaces, gases, and flames in the fire environment result in emission of electromagnetic waves predominantly in the infrared and visible portion of the spectrum.
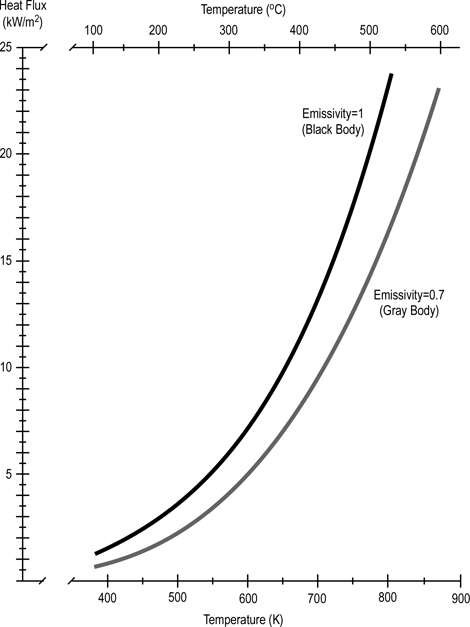
Stefan-Boltzmann Law: The amount of energy per square meter per second that is emitted by a black body is related to the fourth power of its Kelvin temperature. As temperature increases, emission of radiant energy increases exponentially.
A black body is a theoretical object that completely absorbs all incoming radiant energy and is also a perfect emitter of radiant energy. Materials encountered in the fire environment do not completely have the characteristics of a black body and may be classified as gray bodies. A gray body absorbs or emits a portion of the radiative flux depending on the emissivity (?).
Emissivity is the relative ability of the surface of a material to emit radiant energy. It is the ratio of energy radiated by a particular material to energy radiated by a black body at the same temperature. Emmisivity of a black body would be 1.0 with the emissivity of actual materials ranging from approximately 0.1 for highly reflective materials (e.g., polished silver) to 0.97 for fairly efficient absorbers and emitters of radiant energy (e.g., carbon particulate).
Emission of radiant energy is measured as heat flux (energy transfer per unit of time over a given surface area). The SI unit of measure is joules/second/square meter or (since a watt is a J/s) watts/square meter (w/m2).
Figure 4. Stefan Boltzmann Law
Electromagnetic radiation spreads out as it moves away from its source. As a result, the intensity of the radiation decreases as distance from the source becomes greater (as illustrated in Figure 5). The simplest example involves a point source of radiation (distance from the source is much greater than the size (e.g. surface area) of the emitter). With a point source, reduction in radiation intensity follows the inverse square law.
Figure 5. Radiation Intensity Decreases With Distance
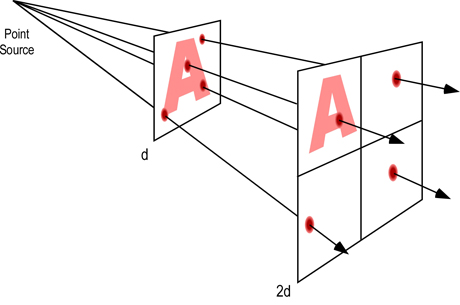
Inverse Square Law: For point sources, intensity of the radiation varies inversely with the square of the distance from the source. Doubling the distance reduces intensity of the radiation by a factor of four (1/4 of its original value).
When radiation is emitted from other than a point source (as it is under fire conditions), variation of the radiation intensity with distance is more complex. If the area of the source is large compared with the distances involved, intensity decreases with distance but does not follow a simple law. As a rough guide, if the distance from the source is greater than about 5 times the dimensions of the source, the inverse square law can be applied.
More to Follow
Subsequent posts in this series will examine physical and chemical changes and the process of combustion.
Ed Hartin, MS, EFO, MIFireE, CFO
Tags: fire behavior, Fire Behavior Training, heat transfer, practical fire dynamics



April 14th, 2010 at 21:32
Great posts on everyday concepts. I can not say I totally understand all of it, but it does make sense. In a fire you have heated gassed that are preheating the compartment by convection, and all the particles within the smoke are radiating heat as they move through the smoke, as they cool they tend to stop moving and stick to a surface, where they conduct the last bit of heat. They also move the surface they landed on closer to being a blackbody.
I had a question though. I seems in a structure fire (and basic weather for that matter) that thermal energy does not move nicely from warmest to coolest. It seems that if a nozzle is sprayed into an area a relative cold spot is made as the heat is absorbed, and a heat increase can be seen at the opposite end of the fire. As if the heat runs away from the cool. I show new guys this I use a great test where I have them hold a hot cast iron pan under a faucet of cold water. The heat from the pan runs up the handle and they drop the pan. This same thing seems to happen with hot gases in a compartment fire.
The only real world example, outside a compart fire, is the wind caused by a cold front blowing in.
This condensing of the heat away from a fire stream that is not flowing enough gpm is what I have believed leads to some rapid fire progression of the years.
I have seen it, I have felt it, can you explain it?
April 15th, 2010 at 07:00
Charles,
Here is my best shot at answering your question:
In discussing heat transfer through convection, you are correct in observing that the fluid mass transfers energy to surfaces through radiation (largely) and conduction (less so). However, you r last sentence “they also move the surface they landed on closer to being a black body” is not entirely correct. Similarity to the theoretical black body is based on emissivity. While emissivity is somewhat dependent on temperature, is also dependent on many other factors such as temperature, surface characteristics, and chemical makeup.
You are correct that things are a bit more complicated in a compartment fire. Temperature gradients are not generally smooth and clearly defined (see Townhouse Fire: Washington, DC-Computer Modeling Part 2). This is due to a variety of factors including natural and forced convection, gravity current, cooling of hot gases as they move away from the fire, mixing of hot gases and cooler air. The energy in the hot gases is dependent on their mass, making mass transfer a significant factor in transfer of energy away from the immediate area of the fire. Application of water in a fog pattern will cool the gases (or surfaces) that it contacts, but also has some potential (particularly if used ineffectively) to move air (and hot gases in the direction opposite the stream). This influences mass transfer (and thus heat transfer) through forced convection.
Cheers,
Ed
April 15th, 2010 at 13:42
Thanks for the reply.
On the one point, about the emissity, what I was thinking is that by covering the surface near the fire with black soot the surfaces emissity would change; bring it closer to the theriortical black body. The carbon covered surface now radiates energy back into the convetion stream and the soot (mass) can go further from the fire before it cools. Though I may be wrong on that.
It makes sense that a water stream, would cause a type of forced convection. I will reread your posts on that point.
I look forward to more posts, and the book for that matter.
take care,
cwilliams